-
Jul 24, 2024Management Guidelines for Whiteflies and CYSDV on Fall Melons in 2024
After Bemisia whiteflies invaded the desert in 1991, production of fall melons became very difficult, and acreage declined in many desert growing areas. With the Section 18 registration of Admire 2F in 1993, growers could effectively control whiteflies again and soon began to profitably produce fall melons in Yuma, and central Arizona. Then in 2007, a new challenge emerged for melon growers when Cucurbit Yellows Stunting Disorder Virus (CYSDV) became established throughout the desert Southwest. It’s taken some time, but economic production of fall melons is back on track in Arizona. In the past several growing seasons, whitefly populations during the fall have been lighter than normal, and CYSDV incidence on fall melons had been held below 20% incidence except for the 2021 season when CYSDV incidence increased dramatically even though whitefly abundance was relatively light (see graph below). This occurred due to abandoned watermelons (~200 acres) in the Tacna area that were left untreated all summer. The watermelon acreage was found 1-5 mile southeast (upwind) of several cantaloupe and honeydew fields and had an abundance of whiteflies and heavily infected with CYSDV. Although the abandoned watermelons had not been irrigated during the summer, heavy monsoon activity in July and August (>3” rain), sustained foliage in the fields and allowed both whitefly adults and CYSDV to build up to unusually high levels. We estimated that the incidence of CYSDV in all the cantaloupe/honeydew fields in the Tacna and Roll areas was greater than 80% at harvest. Melon yields were significantly impacted, and several fields were not harvested. These results stress the importance of cultural management of whiteflies exemplified by results of the fall 2022 and 2023 season (as well as the 2015-2020 growing seasons) in the Tacna/Roll growing area. To download the most recent information for whitefly and CYSDV management in fall melons go to 2024 Guidelines for Whitefly / CYSDV Management on Fall Melons.
The epidemiology of CYSDV is a complex relationship between the virus, the adult whitefly vector, and our local melon cropping system. It has been our goal over the past 17 years to understand this pathosystem and develop practical approaches for reducing CYSDV impact on fall melon production. We continue to develop new information on cultural and chemical control tactics for adult whiteflies (see 2024 Guidelines). Our knowledge to date suggests that fall melons produced near cotton, alfalfa or near areas where spring melons were recently produced are at the highest risk of infection. When practical, growers should isolate fall melon plantings as far away as possible from these sources of whiteflies and CYSDV. Growers forced to plant fall melons near these crops should be vigilant in minimizing adult whitefly infestation levels with insecticides during pre-bloom growth stages. This can be achieved with at-planting and side-dress applications of soil insecticide (Venom, Scorpion, Sivanto) and well-timed applications of foliar sprays including PQZ and Sefina, feeding disruptors with a high degree of bee safety. Based on local research, both products will provide excellent adult knockdown and CYSDV suppression when used in a fall management program. Transform, Assail, Minecto Pro and Exirel are also labeled for use on melons and provide rotational alternatives for adult knockdown. Bottom line: cost-effective chemical tools are available, but minimizing whitefly abundance in the surrounding crop landscape (e.g., cotton, volunteer melons) is critical for them to be most effective.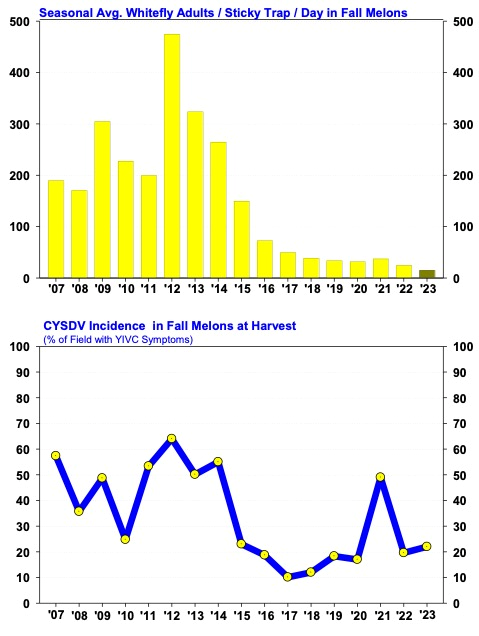 To contact John Palumbo go to: jpalumbo@ag.Arizona.edu
To contact John Palumbo go to: jpalumbo@ag.Arizona.edu















