Feb 9, 2022
High Fertilizer Costs – Update and Background
Throughout the desert Southwest and across the nation, we have continued to experience high prices for fertilizers used in crop production agriculture. Accordingly, there are a lot of concerns and many anecdotes being passed around in an effort to understand the causes. Following a review of the current situation, the conditions that have created these high fertilizer prices, and the prospects for the future; there are many factors involved.
What we are experiencing is illustrated nicely in Figure 1, which describes the pattern of anhydrous ammonia (NH3) costs since September 2008. A very similar story is told in review of the costs for urea (46-0-0), diammonium phosphate (DAP, 18-46-0), and most other fertilizer materials. The high price trends we are now seeing began in September 2020.
Fertilizer prices also experienced a rapid increase in 2008 when nitrogen (N) fertilizer prices increased 32%, phosphate 93%, and potash 100%. Prices then dropped to pre-2007 levels by the end of 2009 and in review the surge was primarily due to high global and national demand and low inventories. The conditions we are now experiencing differ from the 2008 situation.
To better understand this current rise in fertilizer prices it is important to recognize that fertilizer is a global commodity and 44% of all fertilizer materials are exported to a different country. Thus, fertilizer production and prices are affected by other countries demanding fertilizer and the transportation rates to get the fertilizer to the final destination are all important factors.
The U.S. is the third-largest producer of fertilizers globally, and we require the importation of N, phosphorus (P), and potassium (K) to fully meet domestic demand. The U.S. fertilizer dealers and producers pay the price defined by the global market and that include the costs for the base fertilizer, other fertilizer materials, and the transportation requirements.
Anhydrous ammonia (NH3) provides a good example of the U.S. production in relation to the rest of the world. In 2020 NH3 was produced at 36 domestic plants and shipped around the country by pipeline, rail, barge, and truck. As of 2018, U.S. ranked second with 11.6% of global in NH3 production. China at 24.6% led in global NH3 production and India was ranked third with 11.3%.
For phosphate fertilizer production, the U.S. ranked 2nd with 9.9% of global production, led by China at 37.7%, and India with 9.8%.
For the mining and processing of potash (K2O) deposits, Canada is the global leader with 31.9% of global production, followed by Belarus with 16.5%, and Russia with 16.1%. The U.S. produces 0.8% of global potash and ranks 11th in the production of the global supply
Thus, the U.S. is not the sole or dominant player in the global fertilizer industry or market. In point of fact, the U.S. share in terms of global use has dropped from 25% in 1961 to 10% in 2018.
Another major factor to consider are the energy requirements for the production and transport of fertilizer materials. Fertilizer production facilities require a large amount of energy to convert the raw chemical materials into their applicable farm-use state. This is very important in terms of N fertilizers.
There are two basic methods of fixing atmospheric diatomic N gas (N2), which is biologically inert and represents 78% of the earth’s atmosphere. The first, is the natural and miraculous process of biological N fixation which converts inert N2gas from the atmosphere into ammonium-N (NH4). The second is the industrial process, which is also amazing, where anhydrous ammonia is produced by the Haber-Bosch process and atmospheric N2 is combined with hydrogen (H) to synthesize the ammonia (NH3). This reaction is not thermodynamically favorable under natural conditions and huge amounts of energy with high temperatures and pressure are required to accomplish the process. In the Haber-Bosch process, natural gas is the H source and it also the energy source for further N fertilizer synthesis.
Energy costs account for 70% to 90% of the fertilizer production variable costs in the synthesis process. For example, 33 million metric British thermal units (MMBtu) per material ton of NH3 are required to make the conversion in the Haber-Bosch process. Natural gas prices have risen dramatically over the past year, especially in Europe where more than a 300% increase has been experienced since March 2021. This has forced many European Union N plants to close.
The aforementioned factors are dominating the increase in fertilizer prices that we are now experiencing. There are also other important factors including supply chain disruptions, trade duties, and geopolitics. These latter factors tend to get a lot of attention in the agricultural communities and the media often exacerbates that impression. But we see from this basic review, that there are many factors at play, and we can also better understand why fertilizer prices are not likely to come down soon.
Reference:
Myers, S. and N. Nigh. 2021. Too Many to Count: Factors Driving Fertilizer Prices Higher and Higher. Farm Bureau. https://www.fb.org/market-intel/too-many-to-count-factors-driving-fertilizer-prices-higher-and-higher
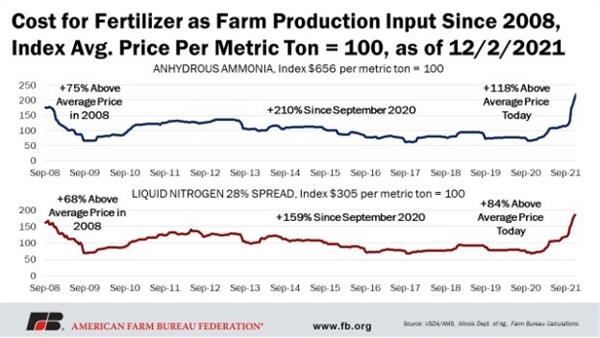
Figure 1. Pattern of anhydrous ammonia (NH3) costs since September 2008.
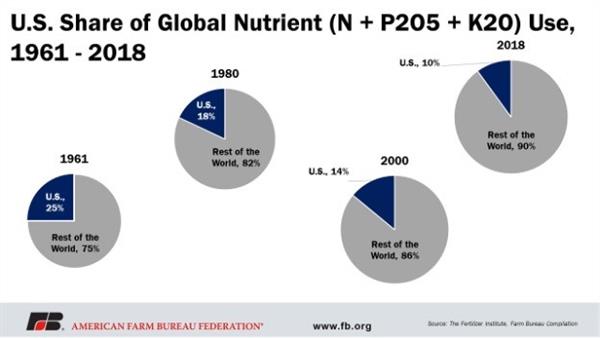
Figure 2. Pattern of U.S. share in the global nutrient market since 1961.
To contact Jeff Silvertooth go to:
silver@ag.arizona.edu