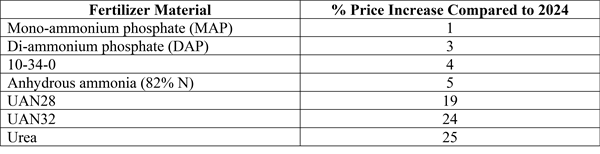
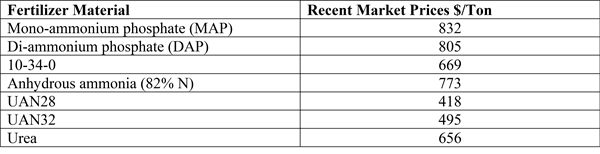
Jan 22, 2025
Bacterial Leaf Spot of Beets (2025)
Bacterial leaf spot is caused by the bacterium, Pseudomonas syringae pv. Aptata. Hosts of the pathogen include Table beet, Sugar beet, Spinach, Swiss chard, Snap bean, Dry bean, Cantaloupe, Pumpkin, Squash, Lettuce, and Pepper.
Significance
Bacterial leaf spot is most commonly found affecting table beet at early stages of growth. This may be because younger plants are more susceptible. It may also be related to the prevalence of cool, wet conditions at the beginning of the cropping season. These conditions are most conducive to infection and disease development. The disease may affect green leaf area in developing seedlings and in severe cases can lead to plant death. Diseased leaves will lead to weakened seedlings which may affect transplant success. Bacterial leaf spot does not directly affect root quality.

Figure 1. Bacterial leaf spot caused by Pseudomonas syringae pv. aptata of table
beet:(left) Small focus of the disease, and (right) 2-6 true leaf stage of plants
affected by the epidemic.
Symptoms
Bacterial leaf spot symptoms are irregular in shape and black to brown in color. The spots may occur across the leaf surface but have a tendency to occur on the leaf edges. Lesions are water-soaked not often accompanied by chlorosis (yellowing). Lesions may initially be small (up to ¼ inch in diameter) but if conditions are conducive may rapidly expand and coalesce but do not cross major veins. The leaf is usually puckered and deformed around the lesions, especially if they occur on the margins (Fig. 2). When the disease is severe, the affected the tissue may also tear giving the appearance of abiotic damage such as hail.

Figure 2. Symptoms of bacterial leaf spot on table beet (cv. Merlin). Note the black
color of the lesions and puckering and deformation of the leaves around the
symptoms.
Bacterial leaf spot symptoms maybe confused with other fungal foliar diseases (e.g. Cercospora and Phoma leaf spots; see complementary fact sheets for these diseases) and insect damage (e.g. thrips). Bacterial leaf spot lesions do not have black pin-head, fungal structures across the lesions as found in Cercospora leaf spot. Phoma leaf spot lesions also have small, black structures within the lesions but found in rings and usually accompanied by an ooze of spores.
Disease Cycle

Figure 3. Schematic diagram of the potential sources of Pseudomonas syringae pv. aptata inoculum which may contribute to Bacterial leaf spot epidemics in table beet.
P. syringae pv. aptata may be introduced to the table beet crop through several ways (Fig. 3). P. syringae pv. aptata is seedborne and infested seed is a common means of pathogen introduction into table beet fields. The pathogen can also be present in the infested crop residues from the previous season as well as the alternative hosts. Alternative crop hosts include typical Chenopods (e.g. spinach, sugar beet, and Swiss chard) but also other non-related species including beans, cucurbits, and lettuce. Cool temperatures between 45-60°F and wet conditions typical of those that occur in early spring in upstate New York are conducive for pathogen infection and disease development. These conditions are similar to those that predispose table beet also to Phoma leaf spot. The pathogen can spread within the field through infested seed and irrigation water.
Disease Management
One of the most critical factors to achieve management of bacterial leaf spot is the use of certified seeds (Fig. 4). Other factors that will also contribute to reducing the initial inoculum and hence risk of disease include: (i) tillage to bury plant residues to promote breakdown, (ii) rotation between host crop species of at least three years; and (iii) drip or furrow irrigation to avoid dispersal of the pathogen through water splash. Currently, little is known of differences in cultivar susceptibility to bacterial leaf spot. Anecdotal reports have described severe epidemics in cvs. Merlin, Boro, and Pablo.

Figure 4. Complementary practices towards the management of Bacterial leaf spot of table beet.
In-season control.
If bacterial leaf spot is severe, applications of copper-based products should be considered to prevent disease spread. There are a range of conventional and OMRI-listed copper-based products available, including: Cueva (copper octanoate; FRAC M1), Badge X2 (copper oxychloride + copper hydroxide; FRAC M1), and Kocide 3000-O (copper hydroxide; FRAC M1). Remember to check the label for rates, re-entry intervals, and pre-harvest intervals applicable to your state and crop. Avoid applying copper-based products on transplants before hardening off, and in high temperatures due to the risk of phytotoxicity.
Source:
https://www.vegetables.cornell.edu/pest-management/disease-factsheets/bacterial-leaf-spot-of-table-beet/