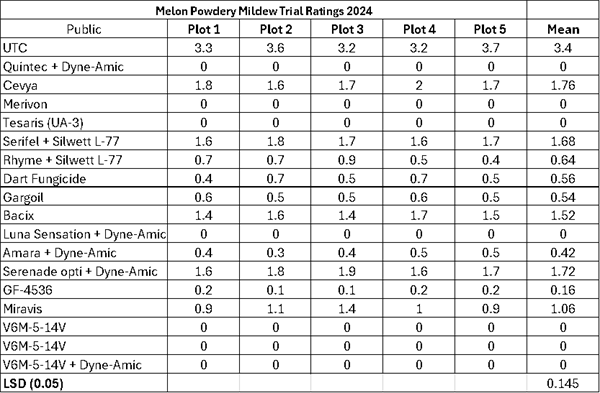
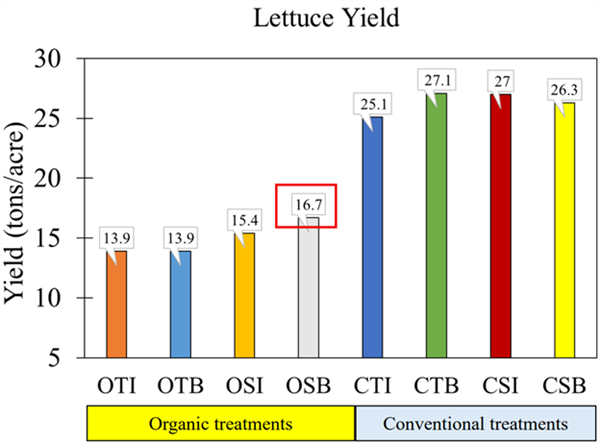
-
Jun 25, 2025Palumbo Thank You Statement
On behalf of the Palumbo family, we would like to extend our deepest gratitude to everyone who made Dr. John Palumbo’s Celebration of Life such a meaningful and memorable event.
A heartfelt thank you to every amazing individual at the farm for your incredible dedication and hard work in planning and executing the celebration with such care and compassion. We are honored that you allowed us to use the farm as the venue to gather, laugh, cry, and share memories and stories of our loved one. We are truly grateful for the time and love that you put into honoring John.
To the generous donors and thoughtful planning committee, we thank you for your kindness, dedication, and outpouring of support. Your generosity has helped create a beautiful space for remembrance, healing, and connection. Your heartfelt speeches touched us deeply, making us both laugh and cry as we remembered and celebrated.
We understand that the loss of John was not only that of a colleague, but a friend and a constant presence in your daily lives. Some absences echo louder than words can express and this is one of them. Even in our sorrow we hold on to the memories, the love, and the moments that will forever be etched into our lives.
It meant so much for all of us to gather and celebrate a life so dearly loved. John would have been honored, and deeply moved, to know his life inspired such a beautiful tribute filled with laughter, stories, and connection.With love,
The Palumbo Family ♥️To contact John Palumbo go to: jpalumbo@ag.Arizona.edu