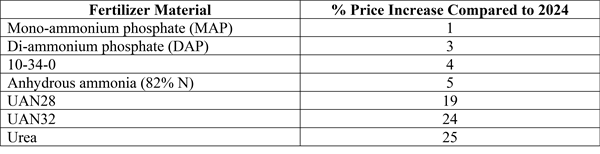
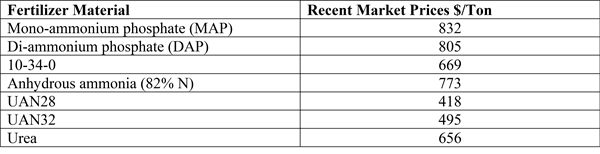
-
Feb 19, 2025Aphids Holding on Strong in Desert Produce
This has been a notable season for aphids in our desert produce crops. Green peach aphids showed up in high numbers in early November and have only increased in abundance during our mild, dry winter so far. At the Yuma Ag Center, numbers in the past few weeks on untreated lettuce were averaging well over 40 nymphs / plant. What’s unusual about this is that dry winters generally result in light aphid pressure. Not this year. We’ve had no rainfall and the temperatures in January and early February have been ideal for green peach aphid. What may also be playing a large role are the high winds we’re had this season. It is well known that winged (alate) aphids migrate into desert produce from outside the area from N/NW wind currents each fall. This year was no different. But wes aw an unusually large peak in trap counts in early November (see graph below). Furthermore, different from most years, counts show that they have been consistently moving into desert produce throughout the winter, peaking at very high numbers last week following heavy winds. Because the desert is very dry, aphids appear to be moving directly into the valleys due to the lack of desert weeds. This has resulted inconsistently high numbers of winged aphids found in our treated and untreated lettuce throughout the winter.
This constant immigration of winged aphids has also affected my insecticide trials. In spring lettuce studies, we’ve observed less efficacy of nymphs than normal. For instance, products like Sequoia, Sivanto and Beleaf normally reduce green peach aphid nymphs by 80-90 % at 7days following application. This year we’re observing 50-60% reductions. I don’t attribute this response to poor application, resistance or experimental error, but rather the continuous influx of winged aphids which immediately begin laying live nymphs after landing on plants. Many of these aphids have not been exposed or intoxicated by the insecticide yet giving the appearance of poor control. Reminds me of whitefly adults onfall melons when they are moving daily. So, if you seem a bit concerned about the level of aphid control you’ve had this winter, consider that it may be mother nature working against you.
One last thing. Foxglove aphids and Lettuce “Red” aphids are beginning to show up. We have found small colonies of both aphid species in our lettuce trials in the past two weeks. I’ve also had reports of Lettuce aphid infesting lettuce in Bard/Winterhaven, Tacna and Yuma Valley. This is later than we’ve seen in the past 2-3 years, but remember these species develop faster in warm weather. So, with daytime high temperatures forecasted to be in the 80’s next week, keep a keen eye out for these aphids. They prefer to colonize the terminals so start there on open lettuce. Foxglove aphids also attack celery, preferring to colonize in the hearts.
For more information on these aphids visit these links:
· Lettuce Aphid on Desert Produce
· Managing Foxglove Aphid on Desert Produce
 To contact John Palumbo go to: jpalumbo@ag.Arizona.edu
To contact John Palumbo go to: jpalumbo@ag.Arizona.edu